Connecting pre-university students with professional science

Contextual Curriculum Connections
A9 Thermal energy transfers, temperature, cooling, insulation
in the context of Antarctic activity
Stories of extreme cold, including challenges of building research station buildings and the importance of insulation, with Valentin Shevtsov

Two objects in thermal contact (that is, with the possibility of thermal energy transfer between them) and with the same temperature are in thermal equilibrium.
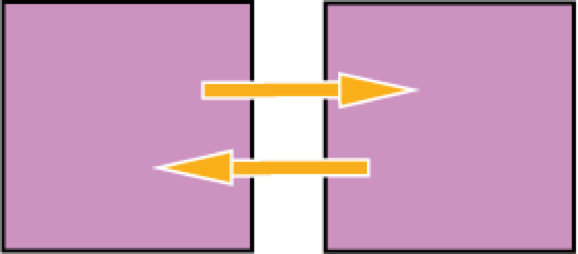
Direct touching is not necessary for thermal contact, because, for example, energy can transfer by exchanging radiation.
Both objects emit and receive energy, but if they have the same temperature there is balance, or equilibrium.

Scientists in research stations like hot coffee.
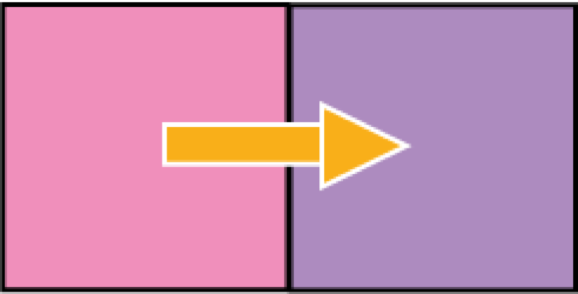
Two objects, like coffee and air, at different temperature are not in thermal equilibrium.
This gives us a possible definition of temperature of any body of material. It is the quantity that determines whether or not an object is in thermal equilibrium with objects (whether solid, liquid, or gas) with which it is in thermal contact.
Note that temperature doesn’t depend on mass, volume, or kind of material (although temperature CHANGE generally does).
If the coffee has a higher temperature than the air then there is thermal energy transfer, The coffee loses energy, and the air gains energy.
The energy lost by the coffee is equal to the energy gained by the air, and other surroundings.
The coffee is much smaller than the surroundings so its CHANGE in temperature is much bigger. We say that its lost energy is dissipated in the surroundings.

Energy transfer occurs until the objects are in thermal equilibrium – that is, until they have the same temperature.
The final equilibrium temperature is intermediate between the two starting temperatures – although if energy is dissipated in large surroundings their temperature rise might be hard to measure.
large initial temperature difference
rapid energy transfer
smaller temperature difference
slower energy transfer
thermal equilibrium: zero temperature difference
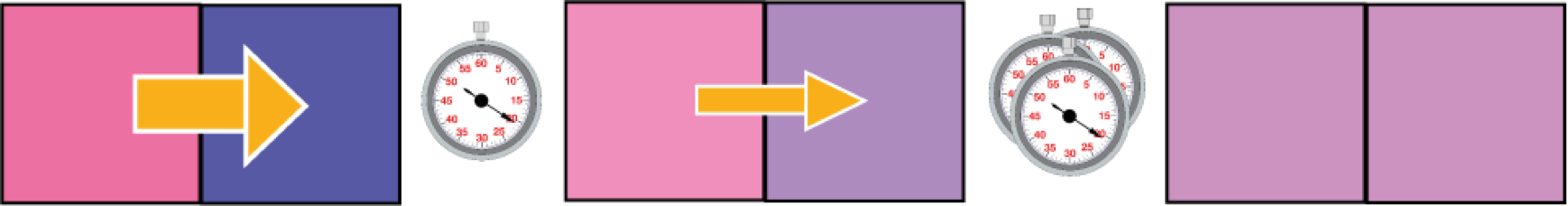
more time
smaller rate of energy transfer
high rate of energy transfer
No energy transfer
In general, energy transfer is faster when temperature difference is higher.
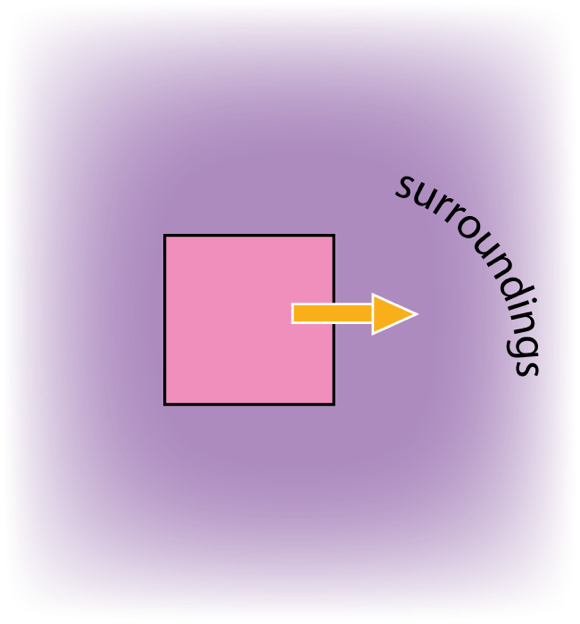
If a body loses energy to large surroundings, so that energy is dissipated, then it will cool to the temperature of the surroundings.
Note that the dissipated energy doesn’t cease to exist. It just becomes very ‘thinly spread’ in the surroundings. The increase of temperature of the surroundings is too small to be measurable.
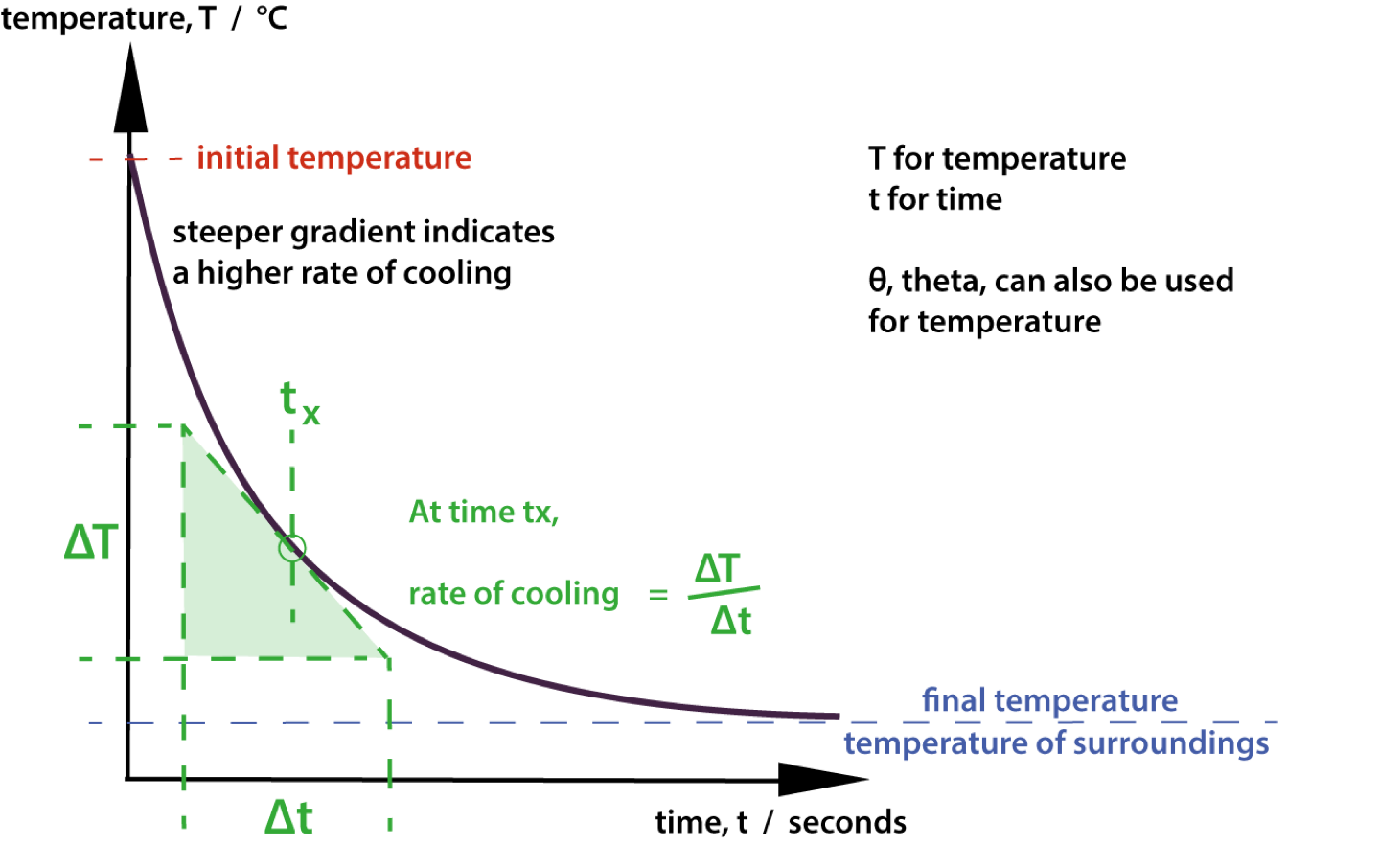
For an object cooling and where the energy it loses is dissipated so that the change in the surroundings is negligible then the following ‘cooling curve’ shows the variation of temperature with time. The gradient indicates the rate of change of temperature at that time.


Energy flows out from a hot machine into surroundings at lower temperature.
Bigger temperature difference produces a faster transfer of energy. (Coffees and hot engines (after they are turned off) cool down fastest when they are hottest relative to the surroundings.)



external energy source
Note that when an engine is running it uses a continuous ‘external’ source of energy (fuel) to replace the energy that it loses to the surroundings as well as to do useful work.
Insulating layer

Slow energy transfer
Insulation reduces the rate of energy transfer so it takes longer for objects to reach equilibrium.
Use of drones in the Antarctic to gather data on snow and to investigate its insulation effects,

In winter the temperature difference between the inside and outside of an Antarctic research station can be much more than 50 °C (50 K),
That tends to produce a very rapid outward flow of energy, but the effect can be reduced with super-high quality insulation of walls, roofs, floors, and windows.
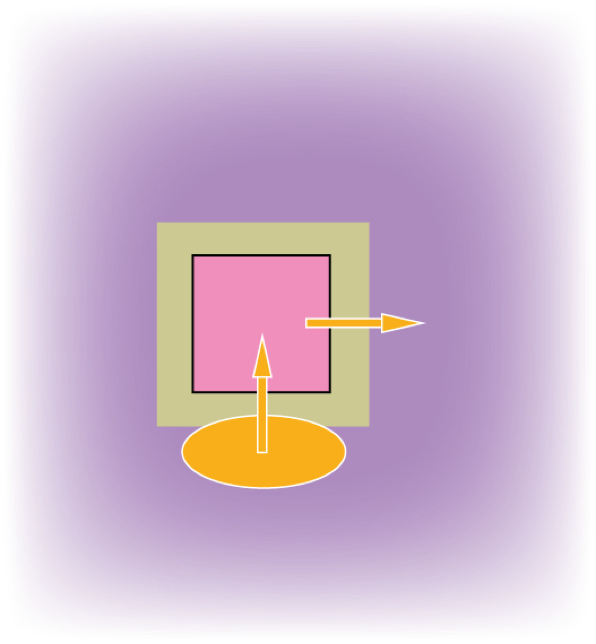
Good insulation can reduce the rate of transfer of energy (represented by arrow width).
However, an external energy source is still needed to replace the outgoing transferred energy.
external energy source
Detail about penguin insulation


A warmer object can sustain its temperature in colder surroundings IF it has a source of energy. (Insulation certainly makes a big difference, but it doesn’t prevent ALL outward thermal flow.)
For penguins and people the source is food.
(C) David Brodie 2025 Note that all external videos and websites remain copyright of the creators
