Connecting pre-university students with professional science

Contextual Curriculum Connections
A10 Measuring thermal energy ins and outs, without and with change of state
in the context of Antarctic ice
A simple introduction to the importance of Antarctic ice.


The iceberg may be in thermal equilibrium with the water and the air – if they are all at the same temperature.
It’s not in thermal equilibrium with the Sun. Ice and Sun don’t have the same temperature and energy is transferring from Sun to ice (even though the Sun is low in the sky).
The ice’s gain in energy could cause its temperature to rise, or if some or all rises above 0 °C then there will be melting.
Water: energy loss or gain without freezing or boiling (without change of state)
When a kilogram of water falls in temperature by one kelvin (1 K) then it loses 4180 J of energy.
For the 1 kg of water to RISE in temperature by 1 K it must GAIN 4180 J.
The value 4180 J/kgK is called the specific heat capacity of water.


Ice: energy loss or gain without melting (without change of state)

When a kilogram of ice falls in temperature by one kelvin (1 K, or one degree Celsius °C) then it loses 2110 joule (2110 J) of energy.
For the 1 kg of ice to RISE in temperature by 1 K it must GAIN 2110 J.
The value 2110 J/kgK is called the specific heat capacity of ice.

With change of state

Unsurprisingly, energy transfers are involved in changes of state, including
solid <—> liquid
Melting won’t happen without a gain of energy.
Freezing won’t happen without a loss of energy.
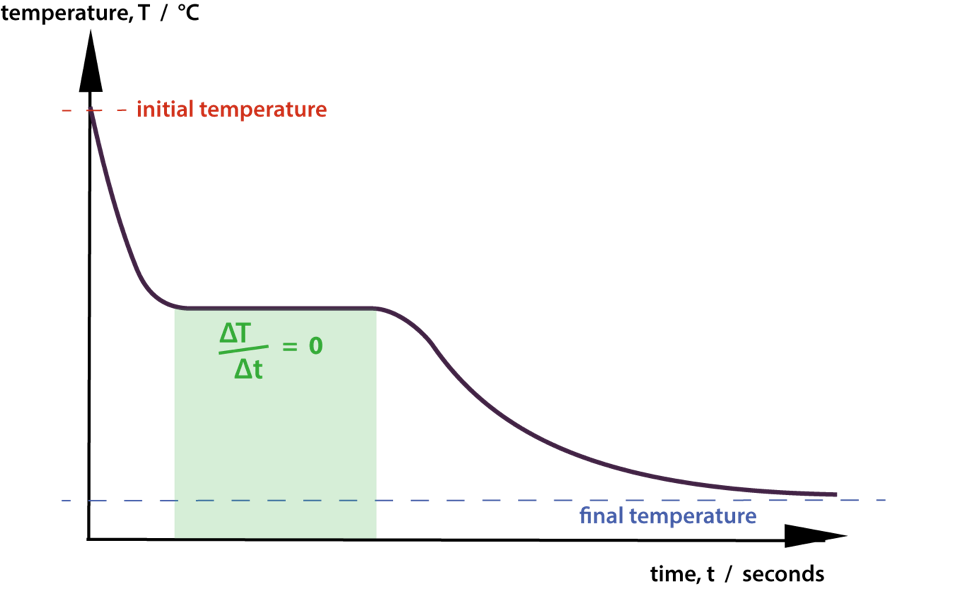
This is a cooling curve for a body losing energy to its surroundings resulting in fall in temperature and in freezing.
During the freezing process the outflow energy continues but it produces solidification only, and temperature stays steady.
For 1 kg of water to change to ice, it must lose 334 kJ of energy.
The specific latent heat of fusion of water is
334 kJ/kg or 334 000 J/kg
A NASA video on quantities of land-based ice melting
Students from Columbia Climate School, Lamont-Doherty Earth Observatory, talk about ice sheets, ice shelves, and sea ice
What happens in the frozen desert of Antarctica is tightly associated with what happens across the planet.

Because of the huge quantity of liquid water on Earth it is very important in sustaining, or not, the temperature of the surrounding air.
The process of melting ice helps to slow down global climate change. There’s a negative feedback link between global average temperature and melting ice. That is, melting ice is a factor that opposes change in temperature.
However, there are many other factors some of which create positive feedbacks. For example, if polar ice reduces the planet will be less reflective of sunlight. Reduction in area of ice is a factor that promotes change in temperature.
Climate science is complex, but all available predictive models tell us that climate change is not only real but is accelerating.

The Earth is a complex body with internal processes, including changes of state,
solid <—> liquid and liquid <—> gas
However, for any object to be stable over time, including having fixed proportions of water as solid, liquid, and gas, then rate of energy input must be the same as rate of energy output.
The Earth’s input is from the Sun. The output is via radiation (infrared, visible light, etc) returned to space.


Greenhouse gases have increased resistance to the outflow. The consequence is that the Earth has an energy imbalance.
Very roughly 1 J of energy more arrives than leaves at each square metre of the planet in every second. That’s an energy imbalance of 1 Js-1m-2 and since 1 Js-1 1 watt, 1 W, we can write that as 1 Wm-2.
One watt is not high power – less than the power of a small LED. But the Earth has a total surface area of about 5 x 1014 m2. So the total imbalance between rate of arrival of energy from the Sun and rate of loss of energy to space is very significant indeed.
The extra greenhouse gases, emitted from human activity in recent generations are the cause of the imbalance. Earth’s climate is slowly (so far) losing its stability.
An introduction to the erosion of the Thwaites Glacier
(C) David Brodie 2025 Note that all external videos and websites remain copyright of the creators
