Connecting pre-university students with professional science

Contextual Curriculum Connections
Antarctic A12 Emission of radiation by atoms
in the context of the southern aurora
A NASAGoddard introduction to the solar cycle and surface activity
A NASA overview of the effects of the solar wind.

The Sun emits a stream of particles, including electrons, protons and some larger ions, and the stream is the solar wind. The Earth’s magnetic field deflects this solar wind away from the planet.
However, especially violent periods of activity on the surface of the Sun produces a more intense solar wind and the stream of particles can then interact with the molecules of the atmosphere. Collisions with atoms and molecules of the gases of the atmosphere excite and even ionise them. Excited atoms or molecules have electrons at higher energy levels. When these electrons lose their extra energy and fall back to lower levels then the gases emit light.
RTE video of the aurora australis (southern lights), above the Chinese Zhongshan Research Station
The aurora australis above the Halley Research Station
The aurora australis above the South Pole, from Timestorm Films

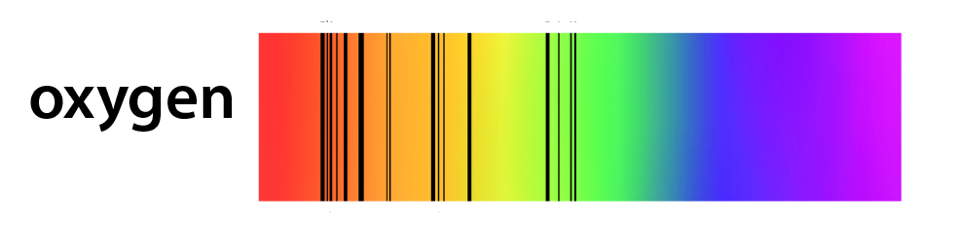
The visible emission spectrum of monatomic oxygen (with single atoms).
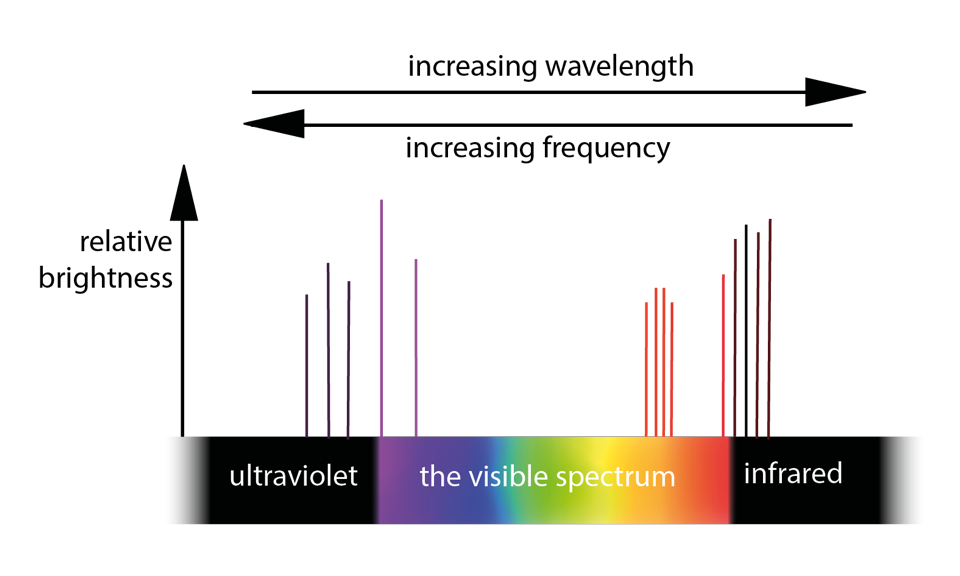
Different elements have different emission spectra.
The emission occurs when atoms with more than the minimum possible energy – excited atoms – lose that extra energy.
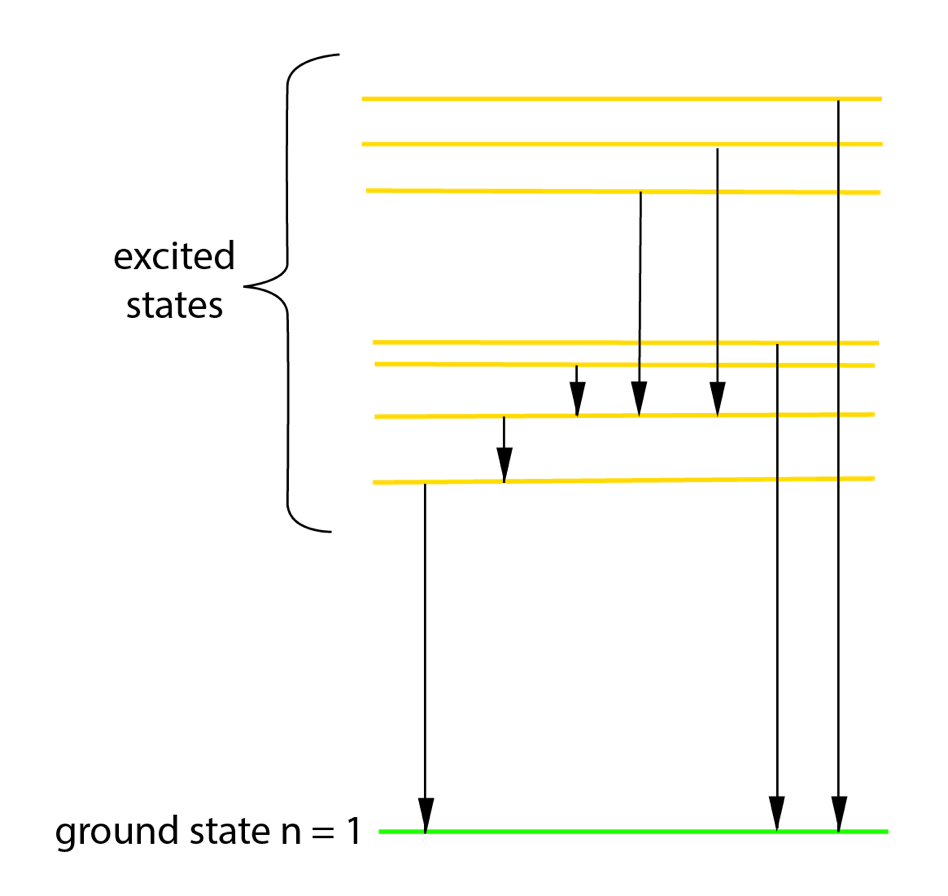
The ‘minimum possible energy’ can be called ground state energy.
Electrons in atoms exist in energy levels. Atoms gain energy, and become excited, when electrons move to higher energy levels.
The diagram shows an example of energy levels (in yellow and green).
The black lines show transitions between levels. These are movements of electrons from higher energy levels, resulting in emission of light.
Longer arrows represent more energy emission – high energy photons which could be in the ultraviolet (UV) range.
Shorter arrows represent smaller energy changes so emission of photons with less energy – possible in the infrared (IR) range.
A single transition event results in emission of a single photon. The energy and frequency of a photon are directly and simple related:
photon energy = hf, where h is Planck’s constant and f is photon light frequency
Planck’s constant = 6.63 x 10-34 JHz-1
So the size of the energy fall directly relates to the size of the frequency, and hence to its colour.

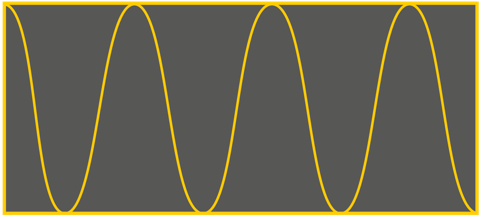
A big ask:
Dear Artist,
Can you create an image of a photon?
Although our vision relies on photons hitting the retinas of our eyes we don’t know what they ‘look like’. Still, it’s part of being human in modern global culture to want to to have some kind of visual representation – a visual model.
The first (upper) is an AI ‘artist’s’ version. It shows a single colour and a nearly-consistent wavelength, wrapped up in a box. The second (lower) is simpler, and with a more clearly uniform wavelength, again inside a ‘packet’.
Of course, gases in the atmosphere exist in diatomic molecules, and this affects their emission spectra.
This diagram shows some stronger emissions from molecular hydrogen, producing a mixture of violet and red light.
The UV and IR emissions are, of course, invisible to the human eye.
What causes an atom or a molecule to become excited or even ionised?
Absorption of incoming photons of light that have precisely the right energy (and frequency) to match a gap between two energy levels can result in an electron jumping up from the lower to the higher of these levels. This means that atoms absorb photons with particular energies (and frequencies) – creating an absorption spectrum with lines with the same colours as the lines of the emission spectrum. So we should expect the absorption spectrum to look like this:
In absorption spectrum, all of the visible colours are present, except those that have frequencies (and photon energies) that match the energy gaps between energy levels.
Excitation and ionisation can also caused by collisions with other very energetic particles. That is what happens when particles of the solar wind hit the gas molecules of out atmosphere.
The aurora australis as seen from the International Space Station – a short video from NASA / Johnson Space
As can be seen in the videos, green is the strongest colour in the auroras, caused by emission of light by oxygen. Pinks, violets and reds match nitrogen emissions.
(C) David Brodie 2025 Note that all external videos and websites remain copyright of the creators
