Connecting pre-university students with professional science

Contextual Curriculum Connections
A7 Masses of atoms and molecules, the atomic mass unit
in the context of testing air and ice
Working with ice cores.
A video from London’s Natural History Museum

Deeply buried ice was deposited many thousands of years ago. Ice cores are obtained by drilling down.
Ancient ice carries information about the ancient climate.
Elena Barbaro from the Ca’Foscari University of Venice tests samples of air and ice cores from polar regions, looking for traces of materials produced by events such as forest fires. In her work she uses mass spectrometry.
Ice cores, especially, hold information about such events in the distant past. They also show how carbon dioxide concentrations have changed over periods of a million years or more.
Elena Barbaro from Ca’Foscari University of Venice explains her work on ice and air samples from the Antarctic and Arctic..
A Canadian video about ice core principles
An instrument created a hundred years ago to measure particle masses is called a mass spectrometer. Now, it can be used to analyse complex organic molecules.
Most exam systems around the world won’t ask you for detail of mass spectrometers. But most of them WILL ask you about key principles: ionisation, magnetic force on moving charges, and masses of atoms
Principles of the mass spectrometer from SCIEX
Measuring masses of atoms and molecules: deflection in a magnetic field
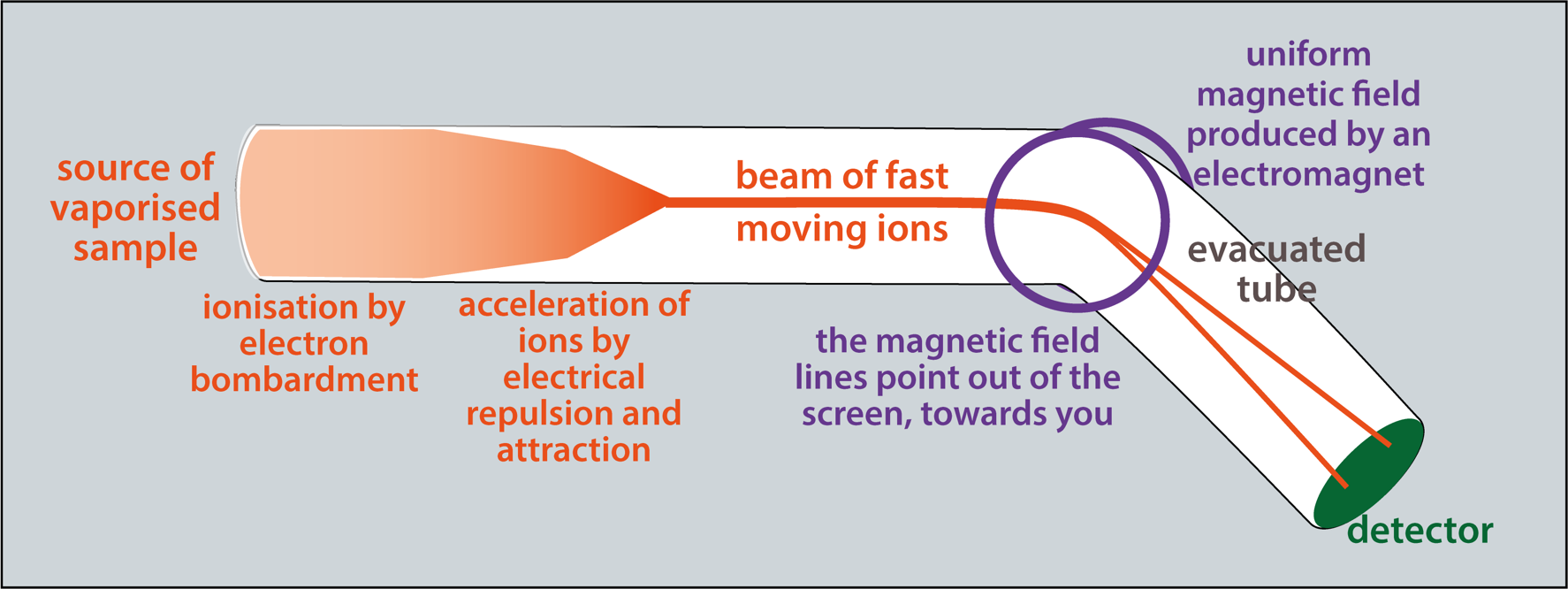
The sample is in the form of vapour and is most commonly bombarded with a stream of electrons (not shown here). Collisions remove electrons from atoms, creating positive ions.
A positively charged plate repels the ions (from left to right in this picture) while negative plates attract them. These plates have holes that the ions can pass through. That produces a beam of ions.
A magnetic field is applied, deflecting the ions. The extent of deflection allows measurement of mass.
See more
Magnetic field and force in the context of auroras
Not yet active.
Internal links coming soon.
NASA video on use of mass spectrometry for analysis of extraterrestrial matter
Creation of a new unit for comparing masses of atoms and molecules
For atoms and molecules, the use of the kilogram, the standard SI unit of mass, is inconvenient, because of the very small numbers.
It makes sense to create a new standard, with which all atomic and molecular masses can be compared.
The isotope carbon-12, 12C, is chosen as the basis for this standard.
1/12 of the mass of a single neutral carbon-12 atom is called 1 atomic mass unit, amu, usually written simply as u. (1 u is also known as 1 Da (1 dalton) although neither of these are official members of the SI system.)
1 u = 1.661 x 10-27 kg.
Mass of 1 carbon atom = 12.0000 u = 12 x 1.661 x 10-27 kg = 12 x 1.661 x 10-24 g
6
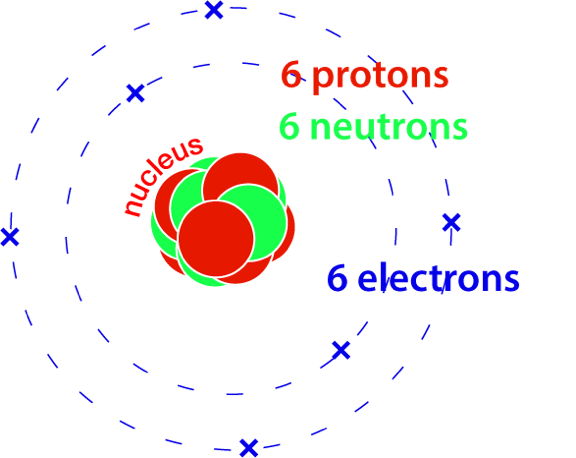
An atom of carbon-12, with twelve particles in its nucleus making up nearly all of its mass.
Carbon-12 is the most abundant isotope of carbon.
See more
Isotopes in the context of analysis of ice cores and ocean sediments
Internal links coming soon.
Ice core bubbles
(C) David Brodie 2025 Note that all external videos and websites remain copyright of the creators
