Connecting pre-university students with professional science

Contextual Curriculum Connections
A8 Counting particles, the mole
in the context of the Antarctic Treaty and the threat of mining

NOT the future of the Antarctic?
In 1959, 12 countries with scientific bases in Antarctica signed an agreement, The Antarctic Treaty. Before that, various countries made claims to part of the continent, and some of those claims still exist, but the terms of the treaty prevent military action or even military bases. They also prevent mining. This means that the only activity is scientific research and a limited level of tourism, and scientists can work alongside colleagues from other countries.
However, the minerals and fossil fuels in the ground are worth a lot of money. Will any country break the treaty and start mining? It hasn’t happened so far.
An Australian account of the recent status of the Antarctic Treaty
A report from Greenpeace International on their campaign for Antarctica to be declared a ‘world park’.

Certainly there is coal underground in the continent.
Plate tectonic movements mean that entire continents can shift around the globe. The Antarctic has not always been a cold place.
Coal – compressed carbohydrate, originally produced by photosynthesis, ended up as mostly simple carbon.
How many atoms are there in 12 grams of carbon-12?
Dividing 12 g by the mass of each atom, also in grams, gives the answer, and it’s (unsurprisingly) a big number.
Based on the definition of the atomic mass unit, u:
mass of 1 carbon-12 atom = 12.0000 u = 12 x 1.661 x 10-24 g.
12 g ÷ (12 x 1.661 x 10-24 g) = 6.002 x 1023
12 g of carbon-12 has 6.002 x 1023 atoms.
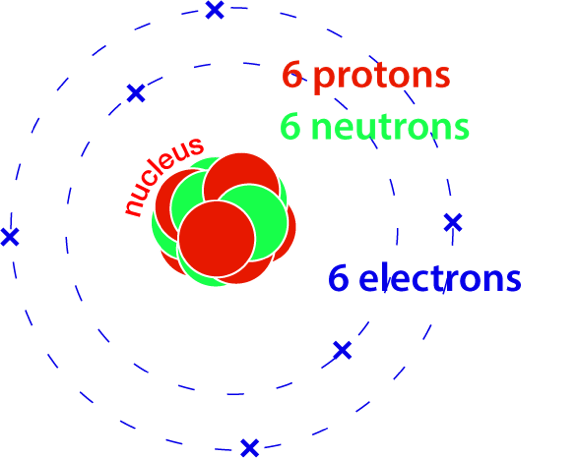
A carbon-12 atom
The mole and Avogadro’s constant
The amount of material with 6.002 x 1023 particles (atoms, molecules, grains of sand) is called a mole, formally abbreviated (very slightly) as a mol.
The number, 6.002 x 1023 , is called Avogadro’s constant, NA.
The mole is important in materials sciences, and is one of the six foundational units of the SI system.

A mol of carbon – 12 g – some black dust on a plate or a large diamond.



Copper oxide, CuO, is an ore of the metal. It is just one of many minerals that some humans might want to exploit in future.
A copper mine. There are no such mines in Antarctica, because every country abides by the Antarctic Treaty (so far).
A visit to You Tolgoi, a huge underground copper mine in Mongolia.
The most abundant isotope of copper is copper-63. It has 63 protons and neutrons making up each nucleus.
The relative* atomic mass of copper-63 is 62.92 u.
That is, mass of 1 copper-63 atom = 62.92 u. =. 62.92 x 1.661 x 10-24 g
62.92 g of copper-63 is 1 mol and has 6.002 x 1023 atoms.
* Relative means relative to 1/12 of the mass of an atom of carbon-12.
I mole of any substance contains 6.022 x 1023 particles.
A single proton has a mass very close to 1 u, at 1.0073 u. The mass of one atom of hydrogen-1 is 1.0078 u.
Mass of 1 hydrogen atom = 1.0073 x 1.661 x 10-24 g
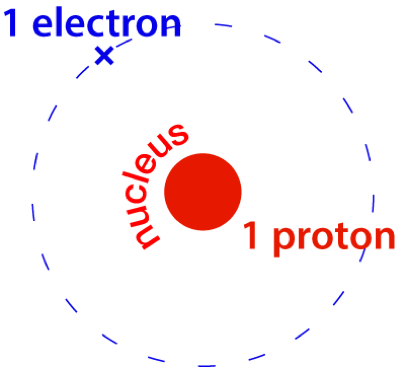
How many atoms are there in 1.0078 g of hydrogen?
We can, likewise, divide the mass 1.0078 g of hydrogen by the mass of each atom, 1.0078 u (after converting this into grams) to find the number of atoms in 1.0078 g of hydrogen.
1.0078 g ÷ (1.0078 x 1.661 x 10-24) g = 6.002 x 1023
What we are doing is dividing a mass in grams by a mass in atomic mass units. We’re going to get the same answer for every substance. It doesn’t depend on the size of the particles. It depends on how many ‘u’ there are in a gram. It’s 6.002 x 1023.
Relative atomic mass, relative molecular mass, molar mass
Hydrogen’s relative atomic mass is 1.0078 u.
However, hydrogen gas exists naturally in molecules, each with two atoms, written as H2.
The relative molecular mass of hydrogen is 2.0157 u.
The molar mass of hydrogen is 2.0157 g mol-1.
How many moles of hydrogen in a weather balloon?
If the ballon has 1400 g and 2 g (approximately) makes up one mole, then a balloon is filled with about 700 mol of the gas.

Watch the video and then work out how many moles of sand there are in the Sahara Desert.
(C) David Brodie 2025 Note that all external videos and websites remain copyright of the creators
